A Master Consolidated Complaint for Individual Claims has been filed in the Cook Medical federal multidistrict litigation (MDL) court in Indianapolis. The Master Complaint will potentially increase efficiency in handling the cases, which make similar allegations.
A Master Consolidated Complaint for Individual Claims has been filed in the Cook Medical federal multidistrict litigation (MDL) court in Indianapolis. The Master Complaint will potentially increase efficiency in handling the cases, which make similar allegations.
The consolidated complaint filed for Cook IVC Filter cases makes the following allegations:
- Cook IVC Filters are associated with an increased risk for serious injury and death resulting from filter tilting, perforation, fracture, breakage, and migration.
- Cook Medical failed to act regarding the known failures and injuries associated with these devices, and failed to warn, concealed, suppressed, omitted, or misrepresented the risks, dangers, defects, and disadvantages of its IVC filters.
- Cook was negligent in its advertising, labeling, promotion, marketing, sales, and distribution of IVC filters, presenting them as safe when the company had reason to know or did know that the devices caused serious injury and death.
- Cook is strictly liable for injuries caused by its IVC filters because the devices are unreasonably dangerous and not accompanied by adequate warnings.
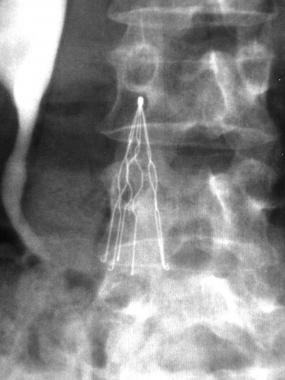
What are IVC Filters?
Inferior vena cava (IVC) filters are used to avert pulmonary embolism (PE), a potentially preventable cause of death in hospitalized and other high-risk patients. The probability of PE increases with the development of deep vein thrombosis (DVT), and the treatment for both PE and DVT is anticoagulation, but in circumstances when blood thinners are not appropriate, an IVC filter is used instead.
The first IVC filters were permanent stainless steel versions, followed by temporary filters fastened to the skin to allow for direct removal without ensnaring. These filters were used in hospitalized patients for a short period of time before they had to be removed.
Currently, the U.S. Food and Drug Administration (FDA) has approved eight optionally retrievable filters, including The Cook Celect and Günther Tulip. Unlike previous temporary filters, optional filters do not need to be tethered to the skin and have a lower rate of infection. They do not have to be removed within a few days, and may be left in place indefinitely, removed when they are no longer needed.
Other IVC Filter Lawsuits News
An Arizona judge denied class certification for Bard IVC filter lawsuits, citing differences among plaintiffs' devices and claims. These lawsuits allege serious injuries from defective IVC filters and insufficient risk warnings.
A new study suggests IVC filters may increase pulmonary embolism risks in traumatic spine injury patients, raising concerns about their safety compared to other preventive treatments.
Studies reveal that Cook Medical’s Celect IVC filters may puncture the vena cava within two months, leading to serious complications and fueling thousands of lawsuits alleging defective design and failure to warn.
A recent study suggests IVC filters may be overused in managed care, often left in place longer than FDA guidelines recommend, raising concerns about patient safety and medical oversight.
Many IVC filters remain in patients indefinitely despite FDA recommendations for removal, raising concerns about serious complications such as fracture, migration, and vena cava perforation.
A new bellwether schedule has been issued in Bard IVC filter litigation, as thousands of lawsuits move forward alleging serious complications linked to the company’s retrievable devices.