A new study published in the Journal of the American Medical Association (JAMA) concludes that use of inferior vena cava (IVC) filters in patients with severe pulmonary embolism (PE) provides no more health benefits than using anticoagulant medications alone, and may actually create more risks to patients.
 In the study, a group of French researchers examined 200 patients who were treated with both blood thinners and IVC filters, and compare them with the same number of patients treated with blood thinner medications only. After a three-month follow-up, twice as many patients with IVC filters were found to have suffered a recurrent pulmonary embolism as those without the filters, leading the researchers to recommend that doctors avoid using IVC filters in patients who could be treated successfully with blood thinners only.
In the study, a group of French researchers examined 200 patients who were treated with both blood thinners and IVC filters, and compare them with the same number of patients treated with blood thinner medications only. After a three-month follow-up, twice as many patients with IVC filters were found to have suffered a recurrent pulmonary embolism as those without the filters, leading the researchers to recommend that doctors avoid using IVC filters in patients who could be treated successfully with blood thinners only.
What are IVC filters?
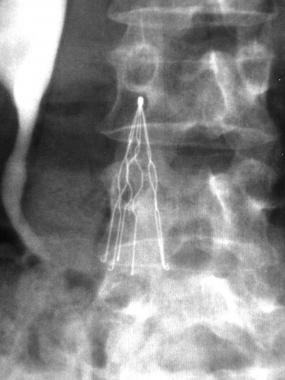
Retrievable IVC filters are small spider-like medical devices that are implanted into the inferior vena cava (the vein that returns deoxygenated blood from the lower legs to the heart) to filter, or catch, blood clots to prevent them from traveling to the lungs, helping to reduce the risk of a PE.
IVC filters are meant to be implanted into the body for a limited amount of time only, and removed when the danger of pulmonary embolism has passed. However, concern has grown that too many IVC filters are not being removed in a timely manner, and instead allowed to remain in the body and expose patients to unnecessary risk, including:
- The progression of deep vein thrombosis (DVT)
- Recurrent PE
- Filter migration
- Filter tilt, break, or embolism
- IVC perforation or occlusion
According to the FDA, the majority of IVC filters that are implanted are not retrieved, and are a frequent choice for doctors and patients, despite their perceived risk of increased fracture, embolism, and IVC wall penetration. In May 2014, the FDA released an updated communication recommending that IVC filters be retrieved 29-54 days after implantation to lower the risks associated with the devices.
Other IVC Filter Lawsuits News
An Arizona judge denied class certification for Bard IVC filter lawsuits, citing differences among plaintiffs' devices and claims. These lawsuits allege serious injuries from defective IVC filters and insufficient risk warnings.
A new study suggests IVC filters may increase pulmonary embolism risks in traumatic spine injury patients, raising concerns about their safety compared to other preventive treatments.
Studies reveal that Cook Medical’s Celect IVC filters may puncture the vena cava within two months, leading to serious complications and fueling thousands of lawsuits alleging defective design and failure to warn.
A recent study suggests IVC filters may be overused in managed care, often left in place longer than FDA guidelines recommend, raising concerns about patient safety and medical oversight.
Many IVC filters remain in patients indefinitely despite FDA recommendations for removal, raising concerns about serious complications such as fracture, migration, and vena cava perforation.
A new bellwether schedule has been issued in Bard IVC filter litigation, as thousands of lawsuits move forward alleging serious complications linked to the company’s retrievable devices.