In 2010, the U.S. Food and Drug Administration (FDA) recommended that inferior vena cava (IVC) filters be removed as soon as the risk of blood clots had passed to avoid complications, and in 2014 the agency repeated this warning. But IVC filters are not always removed once risk subsides and manufacturers are the subject of a rising number of product liability lawsuits alleging serious harm and death caused by these devices.
• On January 17, a Washington man who was implanted with the C.R. Bard Recovery and later with a Meridian IVC filter in 2014 filed a lawsuit alleging injury by the devices. Bard no longer manufactures or sells the Recovery or Meridian IVC filters. C.R. Bard cases are consolidated in multidistrict litigation for pre-trial proceedings in MDL 2641 in U.S. District Court for the District of Arizona, and bellwether trials are expected in 2017.
• A California woman who was implanted with the Cook Günther Tulip IVC filter on October 2, 2007 at Stanford Hospital filed a lawsuit against Cook Medical on January 12. The case will be consolidated with over 1,370 other IVC filter cases pending against Cook Medical in MDL 2570 in the U.S. District Court for the Southern District of Indiana.
• Three class action IVC filter lawsuits were filed in three separate Canadian provinces in 2016: Quebec, Ontario, and British Columbia. A warning issued by Health Canada lists 12 retrievable and permanent IVC filters made by six different manufacturers and says many of the reported complications with them have occurred with filters implanted longer than 30 days.
What are IVC Filters?
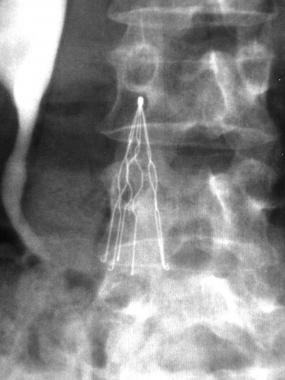
IVC filters are small spider-like devices that are implanted into the vena cava to filter, or catch, blood clots that have broken free to prevent them from reaching the lungs and causing a pulmonary embolism. IVC filters are designed to be implanted in the body for a limited amount of time and removed when the danger of pulmonary embolism has passed; however, concern has grown that too many IVC filters are not being removed in a timely manner and instead allowed to remain in the body and expose patients to unnecessary risk.
Other IVC Filter Lawsuits News
An Arizona judge denied class certification for Bard IVC filter lawsuits, citing differences among plaintiffs' devices and claims. These lawsuits allege serious injuries from defective IVC filters and insufficient risk warnings.
A new study suggests IVC filters may increase pulmonary embolism risks in traumatic spine injury patients, raising concerns about their safety compared to other preventive treatments.
Studies reveal that Cook Medical’s Celect IVC filters may puncture the vena cava within two months, leading to serious complications and fueling thousands of lawsuits alleging defective design and failure to warn.
A recent study suggests IVC filters may be overused in managed care, often left in place longer than FDA guidelines recommend, raising concerns about patient safety and medical oversight.
Many IVC filters remain in patients indefinitely despite FDA recommendations for removal, raising concerns about serious complications such as fracture, migration, and vena cava perforation.
A new bellwether schedule has been issued in Bard IVC filter litigation, as thousands of lawsuits move forward alleging serious complications linked to the company’s retrievable devices.