The C.R. Bard Recovery IVC filter, a medical device intended to prevent blood clots, has instead been associated with further hospitalization, more risky procedures, and 27 deaths over the course of a decade, according to a two-part investigative report aired recently on the NBC Nightly News:
Did Blood-Clot Filter Used on Thousands of Americans Have Fatal Flaw?
Did Forged Signature Clear Way for Dangerous Blood-Clot Filter?
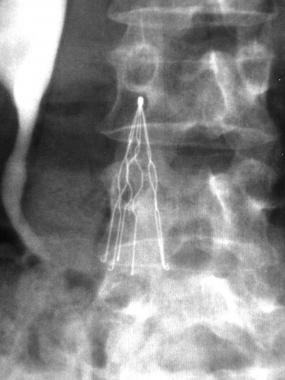
The IVC filter is a metal spider-like implant placed in the main artery beneath the heart and lungs to protect them from blood clots, but numerous patients implanted with the filters may be at risk for serious injuries due to faulty filters that have:
- Punctured veins
- Fractured
- Perforated
- Migrated to other areas of the body and cause other medical problems
- Caused the need for open heart surgery and surgical retrieval
NBC News highlighted several instances where problems with the Recovery IVC filter had severe and  sometimes even fatal consequences.
sometimes even fatal consequences.
Dodi Froehlich, who was the recipient of an IVC filter after she sustained severe injuries in a 2004 car accident that put her at a high risk for blood clots. Four months after the filter was implanted, Froelich developed a severe headache and passed out. Testing revealed that a piece of the IVC filter had broken off and pierced her heart, and she was forced to have emergency open-heart surgery to save her life.
Not all Recovery IVC filter recipients were that fortunate. Gloria Adams needed an IVC filter after a brain aneurysm in 2004. According to her son, Kevin Keech, everything was fine when she was discharged from the hospital, but a week later she was dead. An autopsy revealed that instead of the filter stopping a clot as it should have, a clot pushed the entire device into Mrs. Adams’s heart and punctured it, causing her death.
Each year, about a quarter of a million blood clot filters are implanted in patients who can’t tolerate blood thinners, most without problems. Eleven companies including C.R. Bard sell them in the U.S., but according to the NBC report, the Recovery raised red flags even during FDA approval due to a lack of safety performance test results, a small clinical trial, and possible fraudulent tactics to help gain clearance.
Device manufacturers’ first responsibility is to the safety of the people using their product – it is up to them to ensure that a medical device is fit for use by the public.
When you’ve been harmed by a medical device, you need legal advice from attorneys who have experience standing up to negligent manufacturers and know how to hold them accountable for their products. We pursue nothing less than full compensation for your injuries, and that includes everything – medical expenses, physical rehabilitation, lost wages and pain and suffering.
Call Childers, Schlueter & Smith today or fill out the form on the page to speak with a lawyer who can advise you on the best course of action to take going forward. Our case evaluations are always free, and are always focused on what’s best for our clients.
Other IVC Filter Lawsuits News
An Arizona judge denied class certification for Bard IVC filter lawsuits, citing differences among plaintiffs' devices and claims. These lawsuits allege serious injuries from defective IVC filters and insufficient risk warnings.
A new study suggests IVC filters may increase pulmonary embolism risks in traumatic spine injury patients, raising concerns about their safety compared to other preventive treatments.
Studies reveal that Cook Medical’s Celect IVC filters may puncture the vena cava within two months, leading to serious complications and fueling thousands of lawsuits alleging defective design and failure to warn.
A recent study suggests IVC filters may be overused in managed care, often left in place longer than FDA guidelines recommend, raising concerns about patient safety and medical oversight.
Many IVC filters remain in patients indefinitely despite FDA recommendations for removal, raising concerns about serious complications such as fracture, migration, and vena cava perforation.
A new bellwether schedule has been issued in Bard IVC filter litigation, as thousands of lawsuits move forward alleging serious complications linked to the company’s retrievable devices.