Patients have filed dozens of lawsuits alleging that the blood clot filters manufactured by Cook Medical, Inc. are connected with some severe side effects, including pierced organs and blood vessels that require surgical intervention. Now the first of these cases is scheduled to begin trial in the fall of 2017.
Patients have filed dozens of lawsuits alleging that the blood clot filters manufactured by Cook Medical, Inc. are connected with some severe side effects, including pierced organs and blood vessels that require surgical intervention. Now the first of these cases is scheduled to begin trial in the fall of 2017.
Cook makes more than 16,000 medical devices including stents, catheter balloons, and IVC filters, which account for annual sales of $2 billion and a global workface of approximately 12,000. The company has thus far refused to settle cases involving the filters, insisting that the devices save lives and the pros of using them outweigh the cons.
What are IVC Filters?
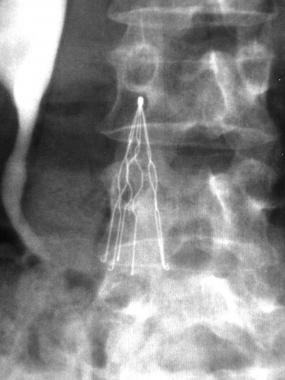
Inferior vena cava (IVC) filters are small devices resembling spiders that are implanted into the vena cava to filter, or catch, blood clots that have broken free to prevent them from reaching the lungs and causing a pulmonary embolism. IVC filters are designed to be implanted in the body for a limited amount of time and removed when the danger of pulmonary embolism has passed; however, concern has grown that too many IVC filters are not being removed in a timely manner and instead allowed to remain in the body and expose patients to unnecessary risk.
Since 2005, the U.S. Food and Drug Administration (FDA) has received more than 900 reports of adverse events connected to IVC filters. According to the FDA, the majority of IVC filters are not retrieved, and their benefits continue to make them a frequent choice for doctors and patients, despite the risk of increased fracture, embolism, and IVC wall penetration.
Cook IVC Filter Lawsuits
The U.S. District Court, Southern District of Indiana has seen a dramatic increase in the number of IVC filter lawsuit filings involving Cook Medical’s Celect IVC filters and Günther Tulip IVC filters, according to court records. The cases generally allege defective design, misrepresentation in marketing, and failure to warn doctors and patients. Besides Cook, other IVC filter manufacturers, including C.R. Bard, are the subject of a rising number of product liability lawsuits alleging serious harm and death caused by these devices.
Other IVC Filter Lawsuits News
An Arizona judge denied class certification for Bard IVC filter lawsuits, citing differences among plaintiffs' devices and claims. These lawsuits allege serious injuries from defective IVC filters and insufficient risk warnings.
A new study suggests IVC filters may increase pulmonary embolism risks in traumatic spine injury patients, raising concerns about their safety compared to other preventive treatments.
Studies reveal that Cook Medical’s Celect IVC filters may puncture the vena cava within two months, leading to serious complications and fueling thousands of lawsuits alleging defective design and failure to warn.
A recent study suggests IVC filters may be overused in managed care, often left in place longer than FDA guidelines recommend, raising concerns about patient safety and medical oversight.
Many IVC filters remain in patients indefinitely despite FDA recommendations for removal, raising concerns about serious complications such as fracture, migration, and vena cava perforation.
A new bellwether schedule has been issued in Bard IVC filter litigation, as thousands of lawsuits move forward alleging serious complications linked to the company’s retrievable devices.