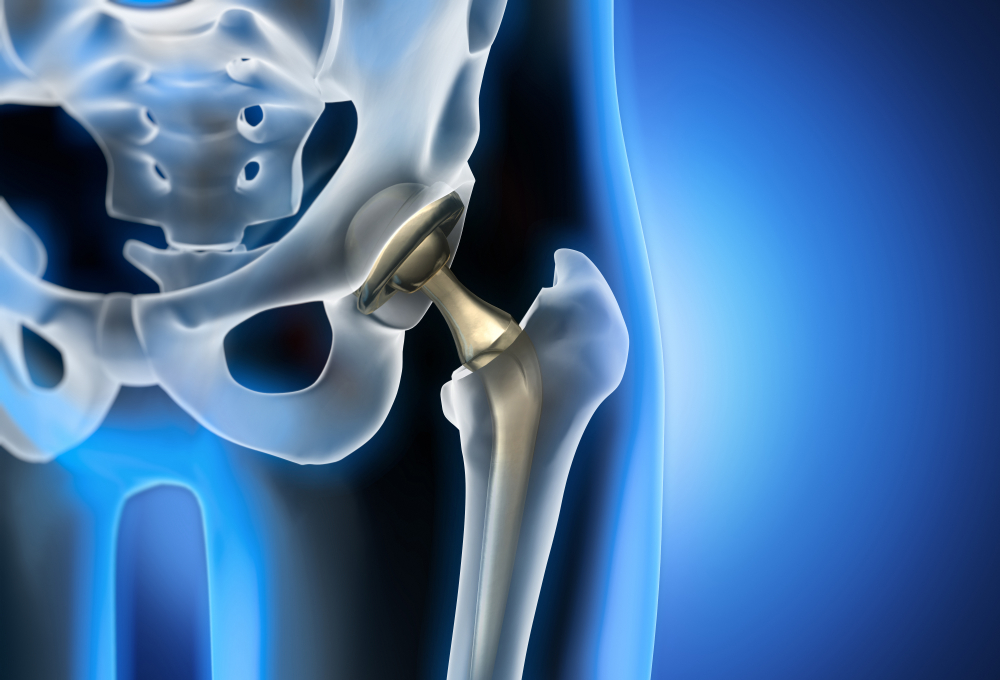
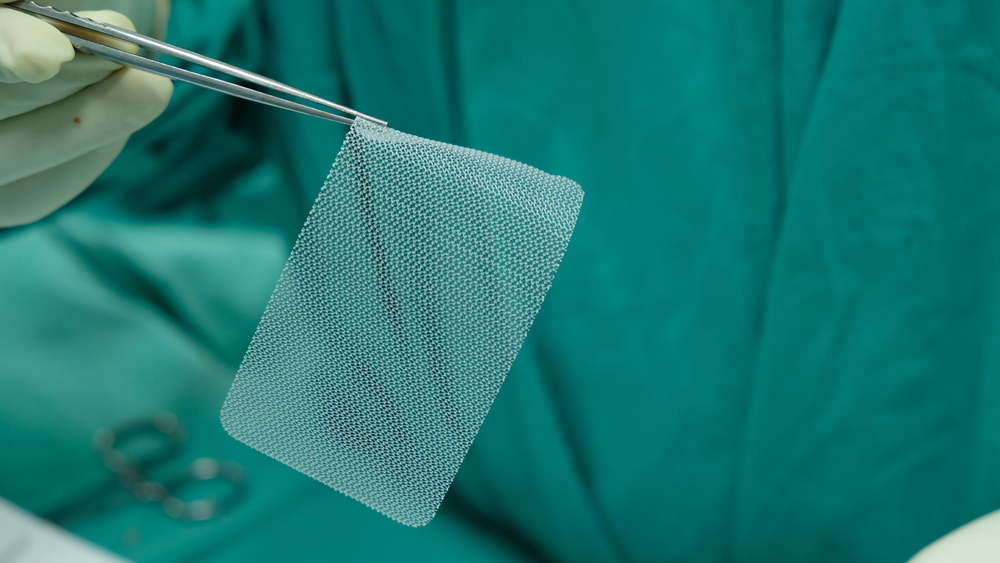
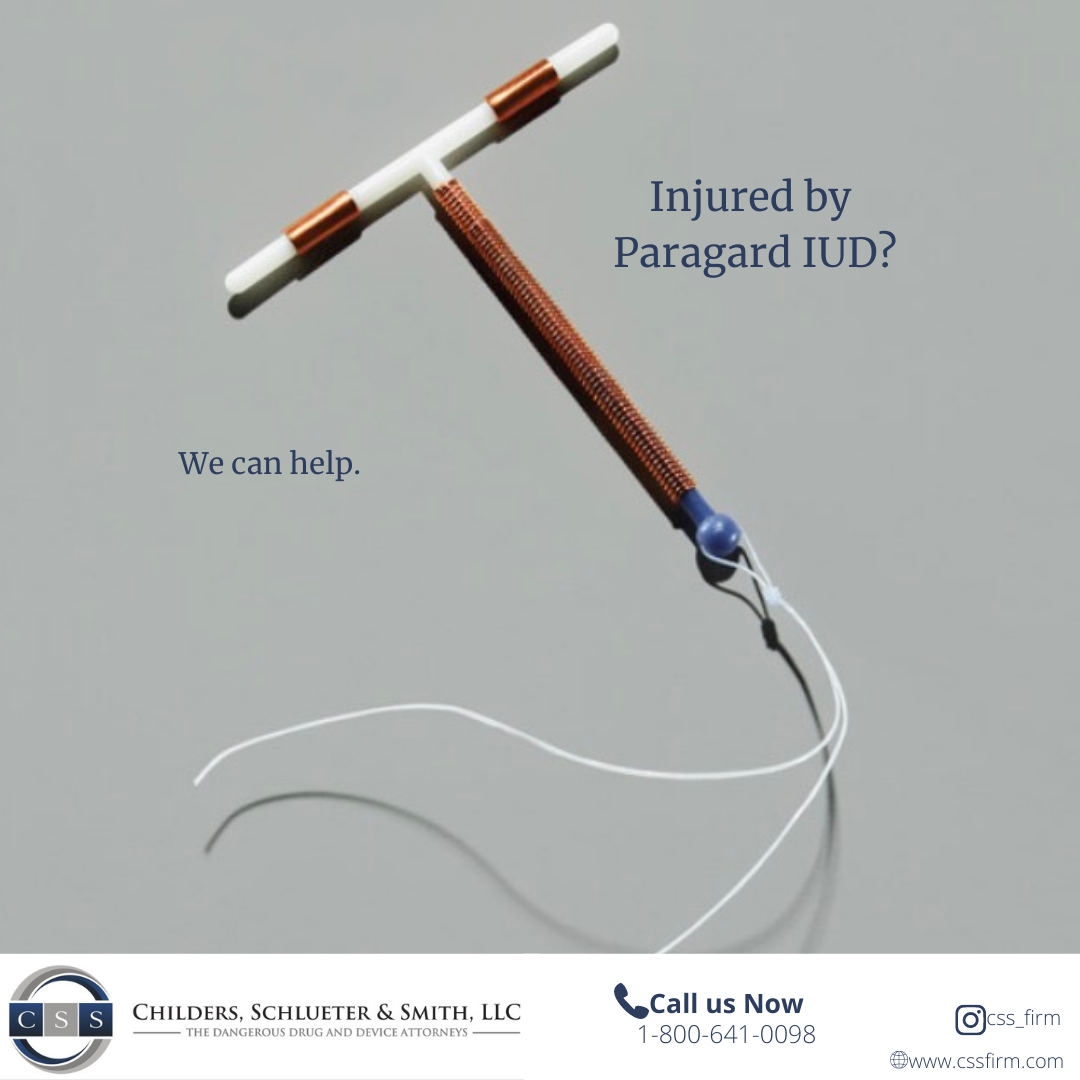
We are pleased to inform everyone there is now an organized and physician backed Joint Registry for those with hip, knee and shoulder implants: www.orthoimplantsite.com
The registry site is the first of its kind to promote awareness and information on joint recalls, issues and potential solutions for patients. It is also designed to help track all hip, knee and shoulder implants which, unbeknownst to most, is currently not required of implant manufacturers by the FDA.
Per the Ortho Implant Recall Site:
If you or a loved one has had hip, knee, shoulder or any kind of joint replacement surgery, the Ortho Implant Recall website is your resource to find out whether or not your implant has been recalled for any reason. Please join our registry and you can rest easy knowing that if the FDA issues a recall or if there is an industry recall on your replacement joint, you will be notified quickly. Your physician has no obligation to notify you so please take advantage of the Ortho Implant Recall registry and join today.
Given all the medical device recalls and claims we have seen over the years, we are happy to see a caring group organize this needed joint registry. We hope it will promote a more informed patient experience and lead to more timely treatment regime if and when needed.
Other Medical Devices News
The FDA warns that Synovo Total Hip Systems, implanted after 2019, pose risks of failure and injury. Patients experiencing symptoms should consult our CSS lawyers to explore legal options for compensation.
Thousands of hernia mesh products have been recalled, but qualifying for a claim requires proof that a defective hernia mesh implant caused your injury.
Damages from a defective hernia mesh implant vary based on each person’s injuries and circumstances. An experienced dangerous medical device attorney can help identify all the losses you may be entitled to recover.
Women nationwide are filing lawsuits after suffering injuries from Paragard IUD breakage—alleging the manufacturer failed to warn of the device’s dangerous design defects.
A voluntary recall affects over 3.5 million Philips Respironics CPAP, BiPAP, and ventilator devices due to foam degradation risks, potentially endangering sleep apnea patients’ health and safety.
Philips Respironics has recalled certain CPAP, BiPAP, and ventilator devices due to degrading sound foam that may release harmful particles and chemicals, posing serious health risks to users.