The U.S. Food and Drug Administration (FDA), is alerting consumers that Meridian Medical Technology is voluntarily recalling 13 lots of Mylan’s EpiPen and EpiPen Jr. Auto-Injector products due to a defect that may cause the devices to fail to activate. Although the number of reported failures is relatively small, EpiPen products containing a defective part that causes the device to fail creates the potential for life-threatening risk when a severe allergic reaction goes untreated.
The U.S. Food and Drug Administration (FDA), is alerting consumers that Meridian Medical Technology is voluntarily recalling 13 lots of Mylan’s EpiPen and EpiPen Jr. Auto-Injector products due to a defect that may cause the devices to fail to activate. Although the number of reported failures is relatively small, EpiPen products containing a defective part that causes the device to fail creates the potential for life-threatening risk when a severe allergic reaction goes untreated.
The recalled lots identified by the FDA are the 0.3 mg and 0.15 mg strengths that were distributed between December 17, 2015 and July 1, 2016, not including the authorized generic version. Patients can receive another EpiPen or a generic version at their pharmacy and in the meantime, Mylan is advising them to continue to carry and use their current EpiPen until they obtain a replace
What are EpiPens?
EpiPens are spring-loaded syringes filled with epinephrine, a potentially life-saving medication used when someone shows signs of a life-threatening allergic reaction to insect bites, foods, drugs, or other substances. Epinephrine acts quickly to improve breathing, stimulate the heart, raise blood pressure, reverse hives, and reduce swelling of the face, lips, and throat.
Pressing the EpiPen into a person’s thigh, which is the prescribed area for administration, causes a needle to penetrate skin and inject epinephrine into muscle. The defective part may require a person to use increased force to activate the needle, or it may prevent the EpiPen from working at all, according to Mylan.
Other Headaches for Mylan Over EpiPen
On April 3, a class-action racketeering lawsuit was filed against Mylan. The lawsuit alleges that the company engaged in an illegal scheme when it dramatically raised the price of the EpiPen over the past decade from $90 to more than $600 for a two pack of the devices. People who either have limited insurance coverage or no insurance at all must pay the list price for the device.
Other Medical Devices News
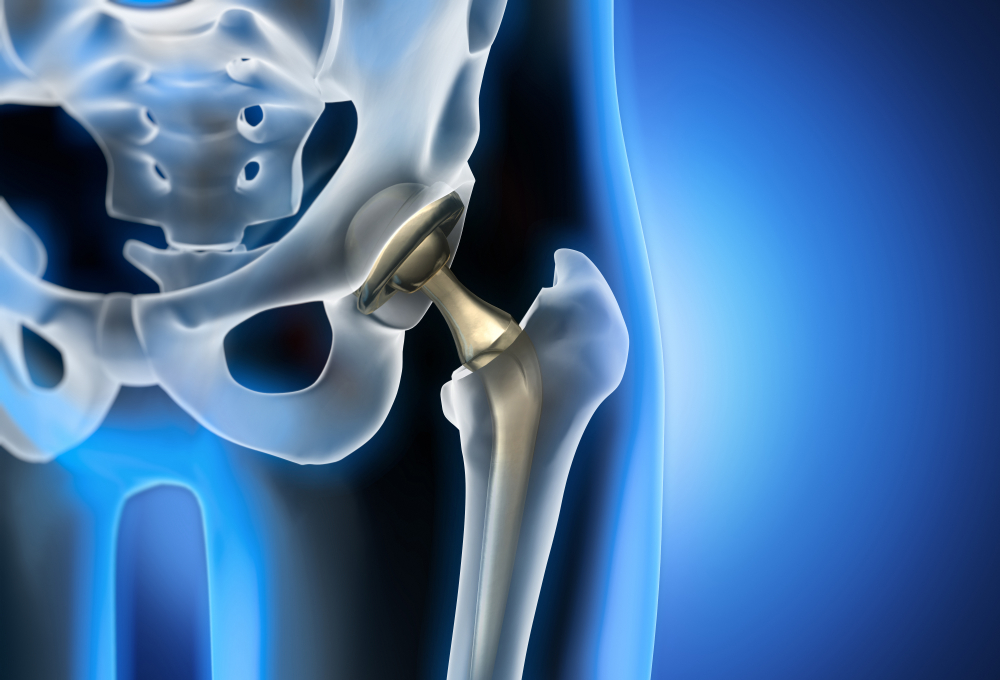
The FDA warns that Synovo Total Hip Systems, implanted after 2019, pose risks of failure and injury. Patients experiencing symptoms should consult our CSS lawyers to explore legal options for compensation.
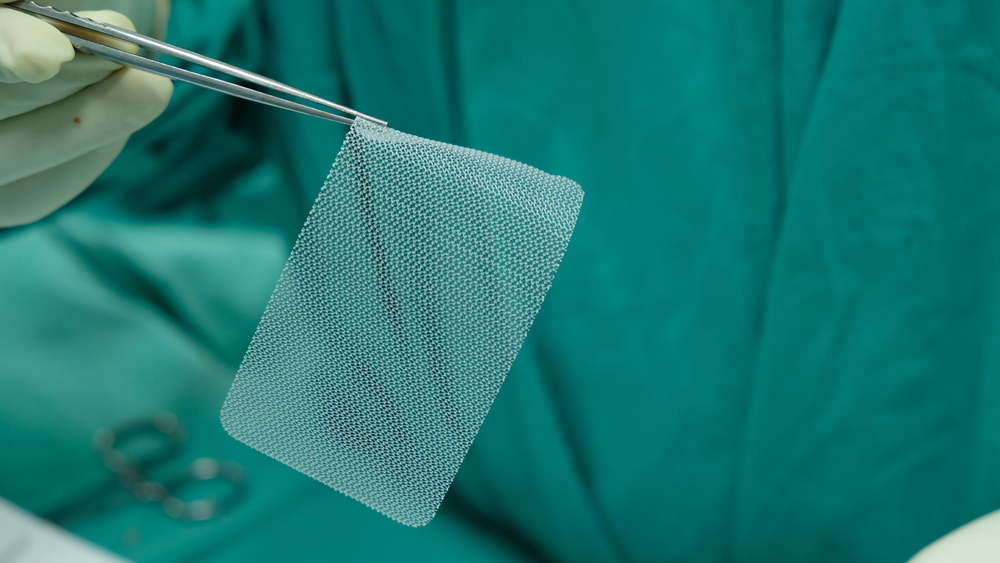
Thousands of hernia mesh products have been recalled, but qualifying for a claim requires proof that a defective hernia mesh implant caused your injury.
Damages from a defective hernia mesh implant vary based on each person’s injuries and circumstances. An experienced dangerous medical device attorney can help identify all the losses you may be entitled to recover.
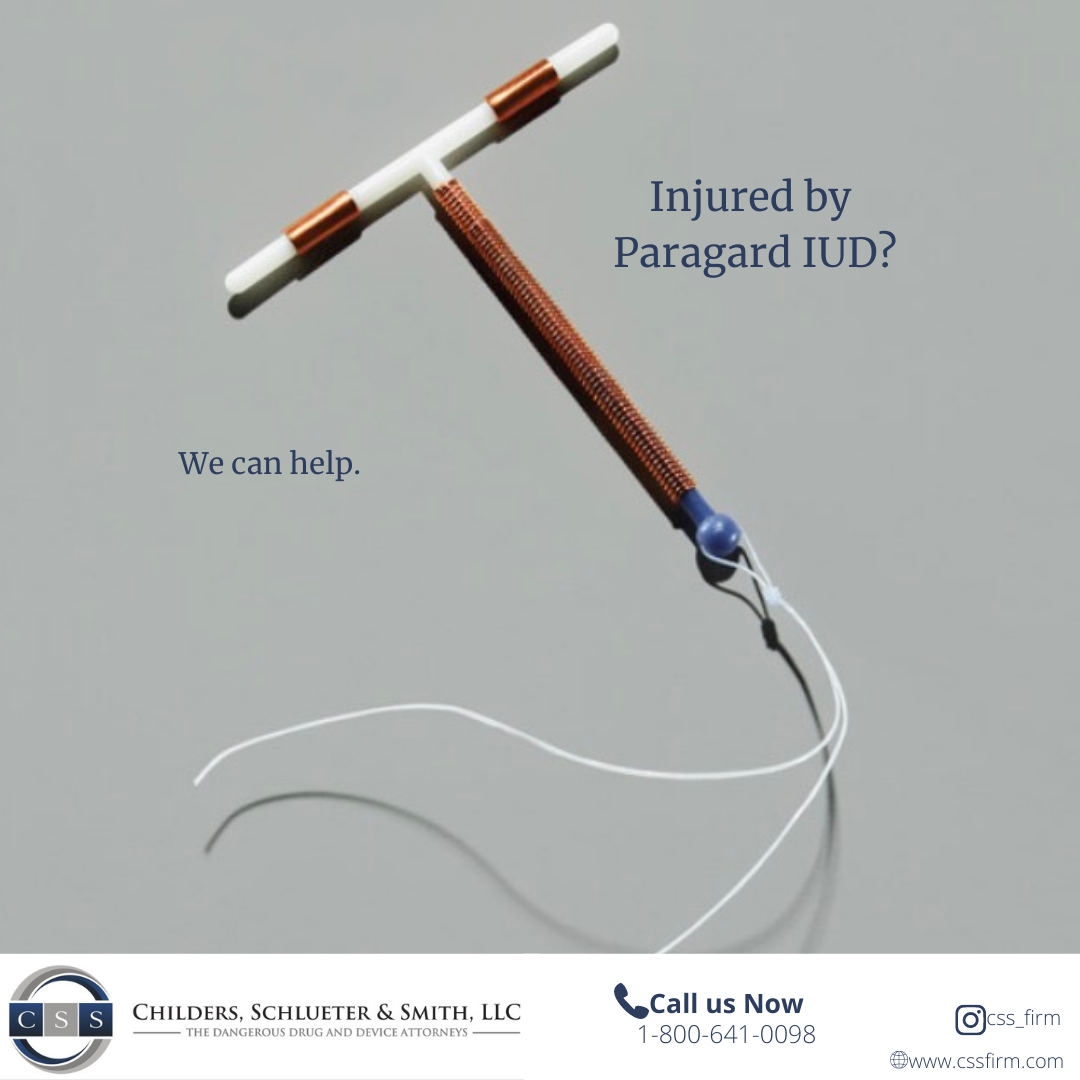
Women nationwide are filing lawsuits after suffering injuries from Paragard IUD breakage—alleging the manufacturer failed to warn of the device’s dangerous design defects.
A voluntary recall affects over 3.5 million Philips Respironics CPAP, BiPAP, and ventilator devices due to foam degradation risks, potentially endangering sleep apnea patients’ health and safety.
Philips Respironics has recalled certain CPAP, BiPAP, and ventilator devices due to degrading sound foam that may release harmful particles and chemicals, posing serious health risks to users.