California Lawsuit Alleges MiniMed Insulin Pump Caused Serious Injuries
A group of seven plaintiffs filed a lawsuit in California Superior Court alleging that Medtronic MiniMed pumps were defective and unfit for the use for which they were intended. The pumps were sold in an unreasonably dangerous condition, causing plaintiffs to suffer injuries. Plaintiffs filed from California, Illinois, Kansas, Nevada, Ohio, and West Virginia in May 2020. The claim has since been moved to the U.S. District Court for the Central District of California.
Six months prior to the lawsuit, Medtronic recalled 322,000 of its MiniMed 600 Series Insulin Pumps because of a manufacturing problem that resulted in either a missing or damaged retainer ring in the affected devices. The ring is designed to secure the insulin cartridge into place in the pump’s reservoir compartment. If the cartridge is not firmly in place, the pump could over/under supply insulin, resulting in either low blood sugar (hypoglycemia) or high blood sugar (hyperglycemia). Severe hyperglycemia can result in unconsciousness, seizure, and possible death.
Specific Medtronic Products in Question

MiniMed 630G Insulin Pump System
The California lawsuit focuses on two Medtronic devices – the Medtronic 670G System and the 630G System, which were approved by the U.S. Food and Drug Administration (FDA) in 2016. The devices simulate functions of the pancreas by dispensing the proper dose of insulin through a catheter connected to a thin cannula placed underneath the skin. The pumps provide continuous delivery to short-acting insulin to type 1 diabetes patients, replacing the need for daily insulin injections.
At the time of the recall and amid 26,421 complaints regarding the device, Medtronic advised customers regarding the defects to check for. The retainer ring on their MiniMed pump should be examined to determine if it is missing, loose, or damaged, and if the reservoir is not properly locking into the pump. With 2,175 reported injuries and one death so far, more lawsuits have been (or are expected to be) filed against the company.
Please Note: This article is intended for educational purposes only. CSS Firm is not currently accepting Metformin/Medtronic claims as research is still developing.
Other Medical Devices News
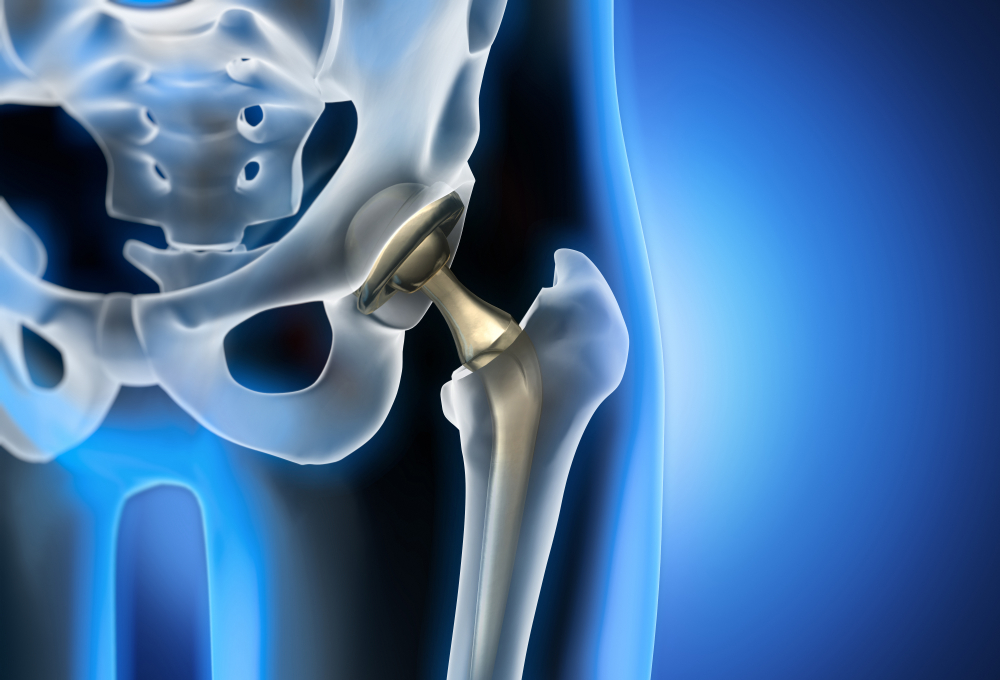
The FDA warns that Synovo Total Hip Systems, implanted after 2019, pose risks of failure and injury. Patients experiencing symptoms should consult our CSS lawyers to explore legal options for compensation.
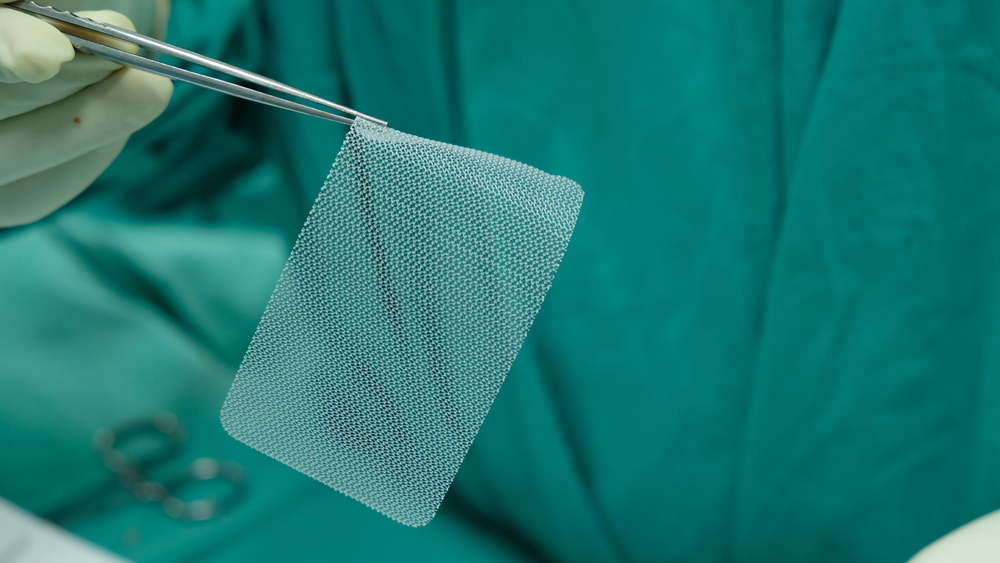
Thousands of hernia mesh products have been recalled, but qualifying for a claim requires proof that a defective hernia mesh implant caused your injury.
Damages from a defective hernia mesh implant vary based on each person’s injuries and circumstances. An experienced dangerous medical device attorney can help identify all the losses you may be entitled to recover.
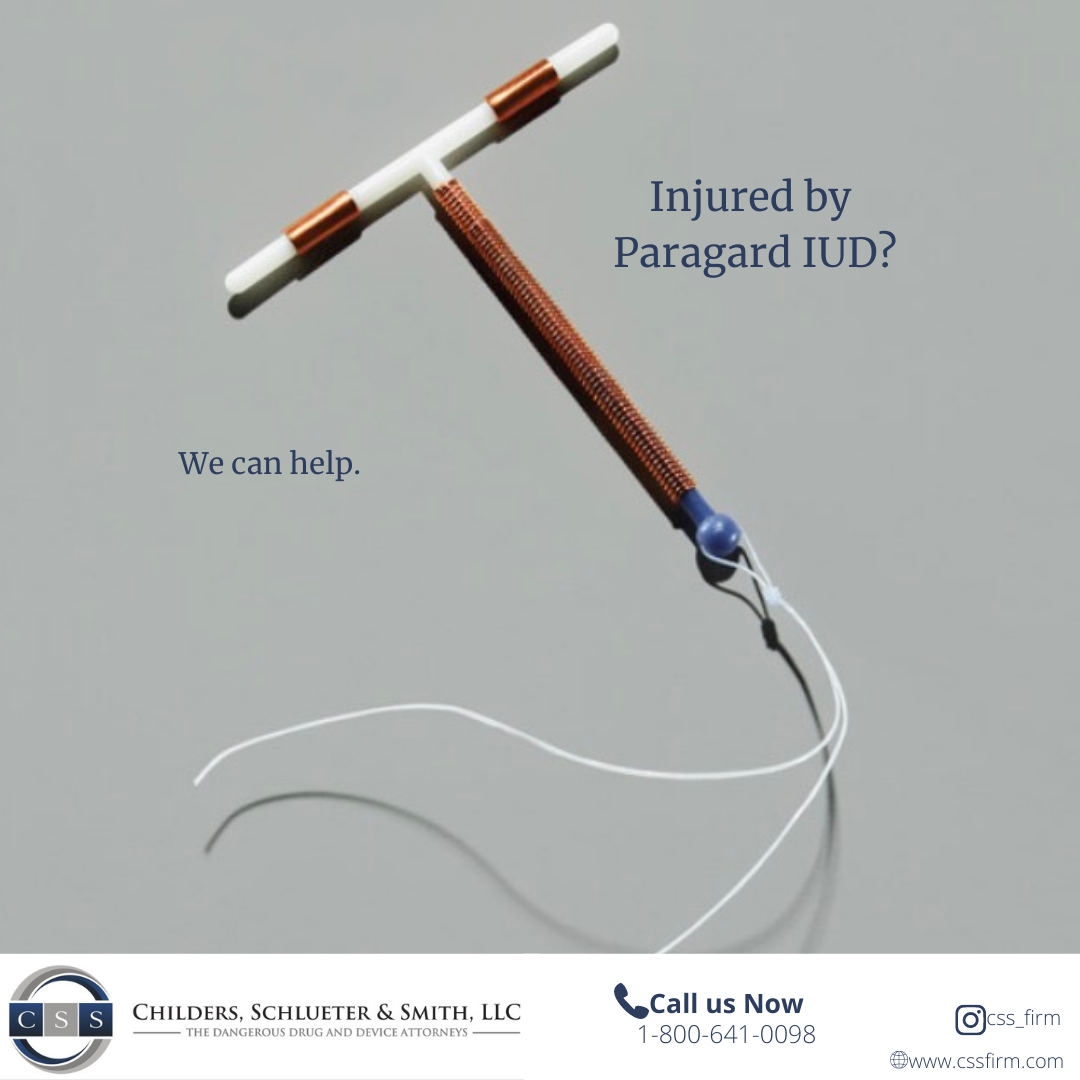
Women nationwide are filing lawsuits after suffering injuries from Paragard IUD breakage—alleging the manufacturer failed to warn of the device’s dangerous design defects.
A voluntary recall affects over 3.5 million Philips Respironics CPAP, BiPAP, and ventilator devices due to foam degradation risks, potentially endangering sleep apnea patients’ health and safety.
Philips Respironics has recalled certain CPAP, BiPAP, and ventilator devices due to degrading sound foam that may release harmful particles and chemicals, posing serious health risks to users.