Please Note: This article is intended for educational purposes only. CSS Firm is not currently accepting Metformin claims at this time.

Metformin Headquarters – Metformin
Medtronic recently recalled more than 322,000 MiniMed insulin pumps with broken or missing retainer rings that led to at least one death, thousands of injuries, and numerous lawsuits. The recall arose after broken or missing retainer rings were observed on the devices. The ring defect that could prevent the insulin cartridge from being locked into place and, as a result, cause people to receive too much or too little insulin.
Insulin overdose can lead to hypoglycemia, a condition in which the blood sugar is lower than normal, leading to lethargy and insulin shock. Underdosage may cause hyperglycemia, which occurs when there are high levels of sugar (glucose) in the blood. Hyperglycemia can lead to a severe condition known as diabetic ketoacidosis (DKA). Without treatment, someone with DKA is in danger of falling into a diabetic coma.
MiniMed Recall Most Serious for Medical Devices
The Class I recall (the most serious type for medical devices) affects all lots of the Model 630G (MMT-1715) manufactured before October 2019 and all lots of Model 670G (MMT-1780) manufactured before August 2019. If you own a MiniMed insulin pump that is part of the recall, you should take the following action:
- Inspect the pump’s retainer ring.
- Stop using the pump immediately if the reservoir cartridge fails to lock into the pump or if the ring is loose, damaged, or missing.
- Communicate with your physician and follow their recommendations for manual insulin injection.
- Contact Medtronic at 877-585-0166 to request a replacement pump.
If the reservoir locks properly, Medtronic advises that you continue to use the pump. If the pump is dropped or damaged accidently, you should check the ring for damage.
According to the FDA, there have been over 26,000 complaints concerning the MiniMed insulin pump. However, despite the fact that the device might cause serious injury or death, it remains approved for use in the U.S.
Other Medical Devices News
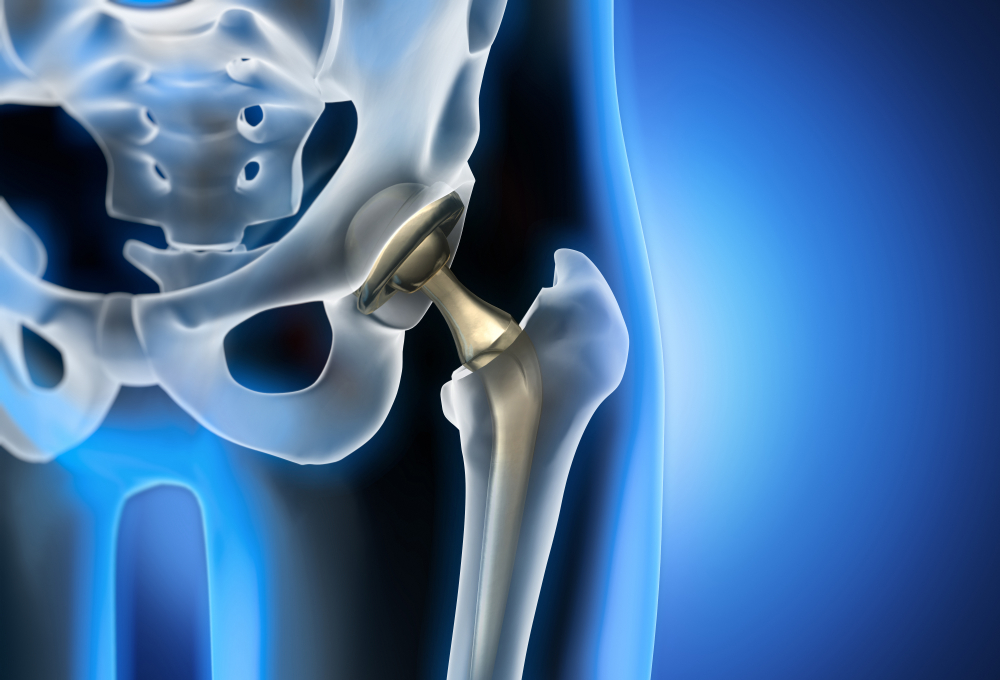
The FDA warns that Synovo Total Hip Systems, implanted after 2019, pose risks of failure and injury. Patients experiencing symptoms should consult our CSS lawyers to explore legal options for compensation.
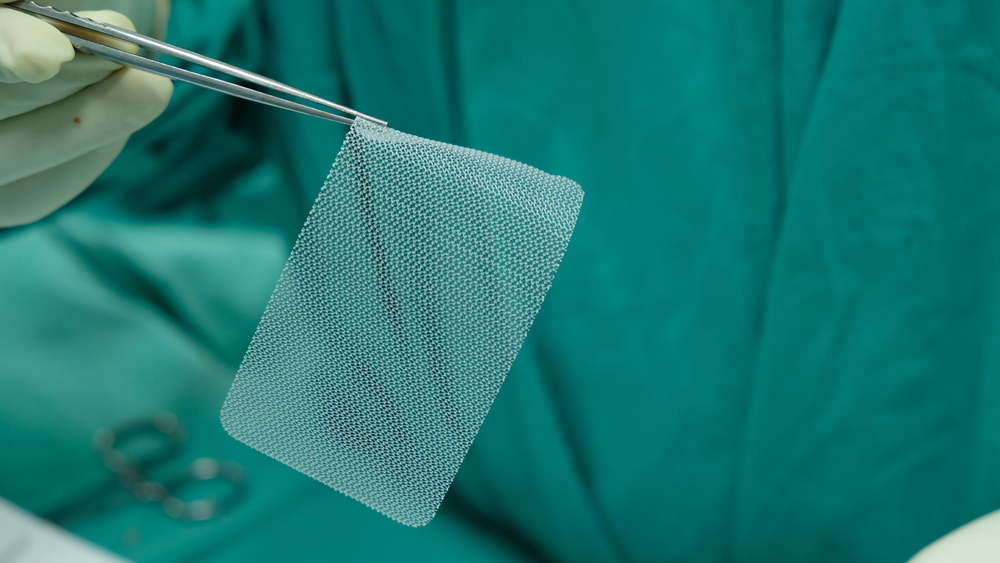
Thousands of hernia mesh products have been recalled, but qualifying for a claim requires proof that a defective hernia mesh implant caused your injury.
Damages from a defective hernia mesh implant vary based on each person’s injuries and circumstances. An experienced dangerous medical device attorney can help identify all the losses you may be entitled to recover.
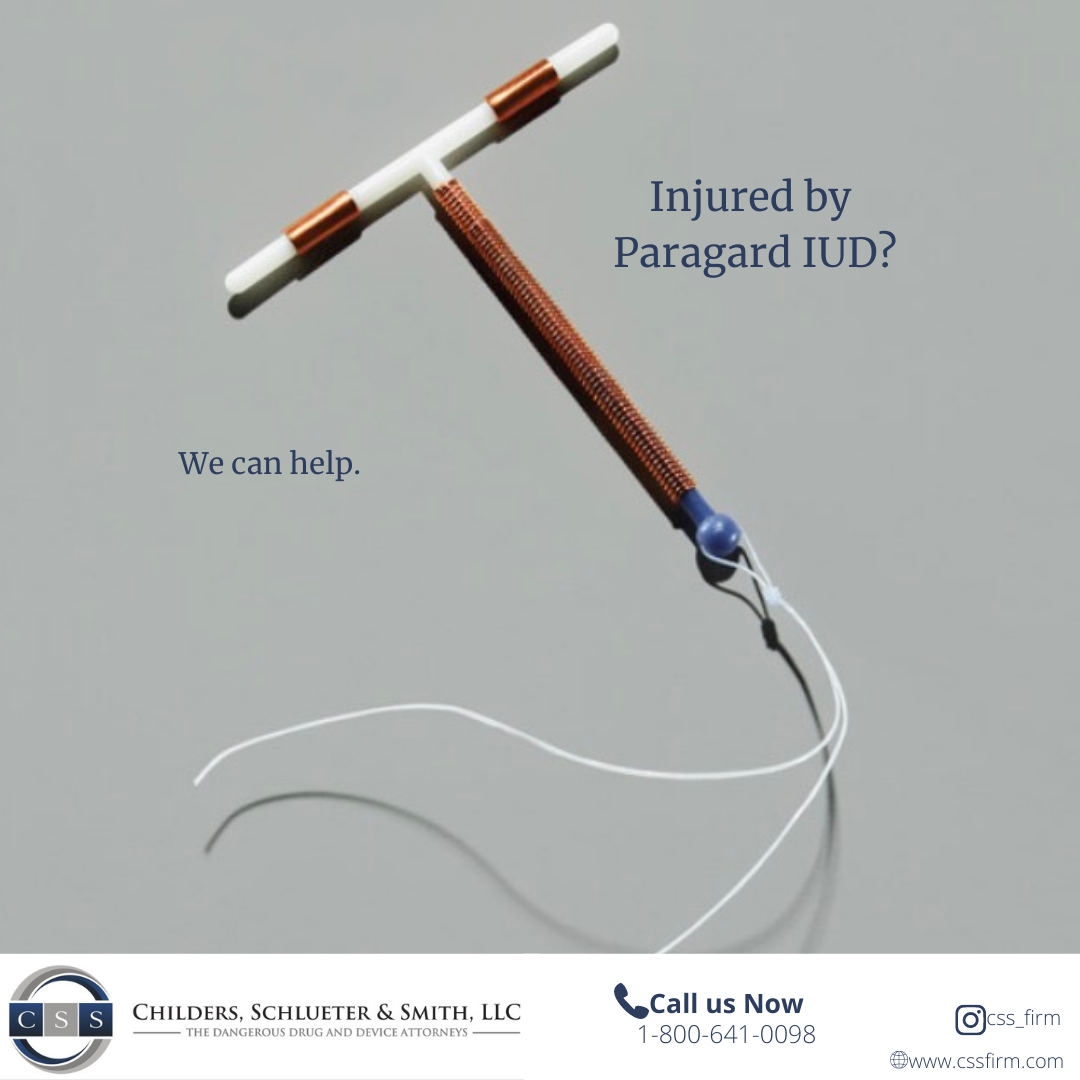
Women nationwide are filing lawsuits after suffering injuries from Paragard IUD breakage—alleging the manufacturer failed to warn of the device’s dangerous design defects.
A voluntary recall affects over 3.5 million Philips Respironics CPAP, BiPAP, and ventilator devices due to foam degradation risks, potentially endangering sleep apnea patients’ health and safety.
Philips Respironics has recalled certain CPAP, BiPAP, and ventilator devices due to degrading sound foam that may release harmful particles and chemicals, posing serious health risks to users.