On December 15, the U.S. Food and Drug Administration (FDA) issued an Urgent Letter to Health Care Providers advising users of the Penumbra JET 7 Reperfusion Catheters with Xtra Flex Technology to stop utilizing the device. On the same day, Penumbra initiated a voluntary recall of the JET 7 with Xtra Flex from the U.S. market due to the device’s susceptibility to distal tip damage during use.
According to Penumbra’s Urgent Voluntary Medical Device Recall Notification, use of the device has been associated to an increased risk of mortality and serious injury in stroke patients. As directed in Penumbra’s Recall Notification, all users should stop using the device and facilities should remove it from their inventory. The company estimates that approximately 30,000 units of the recalled device have been distributed, and of those units, 17 events involving patient injury and 14 events involving patient death have been identified.

Penumbra JET 7 Reperfusion Catheter
According to the FDA, device failures reported include “ballooning, expansion, rupture, breakage or complete separation, and exposure of internal support coils near the distal tip region of the JET 7 XTRA Flex catheter,” leading to various serious injuries like blood vessel damage, hemorrhage, and cerebral infarction.
What is the Device Used For?
The 2018 Penumbra JET 7 Reperfusion Catheter with Xtra Flex Technology is a medical device intended to restore blood flow by eliminating clots via continuous aspiration in patients experiencing an acute ischemic stroke. The device enables physicians to use minimally invasive aspiration to remove stroke-causing blood clots from the brain safely and effectively.
The FDA has received over 200 medical device reports (MDRs) associated with the JET 7 XTRA FLEX including malfunctions, serious injuries, and 14 patient deaths associated with use of the device. Those who experienced major complications or death as a result of the use of a faulty Penumbra JET 7 Reperfusion Catheter may be entitled to monetary compensation for their damages.
Please Note: This article is intended for educational purposes only. CSS Firm is not currently accepting Penumbra JET 7 Catheter claims at this time as research is still developing. Check back for updates.
Other Medical Devices News
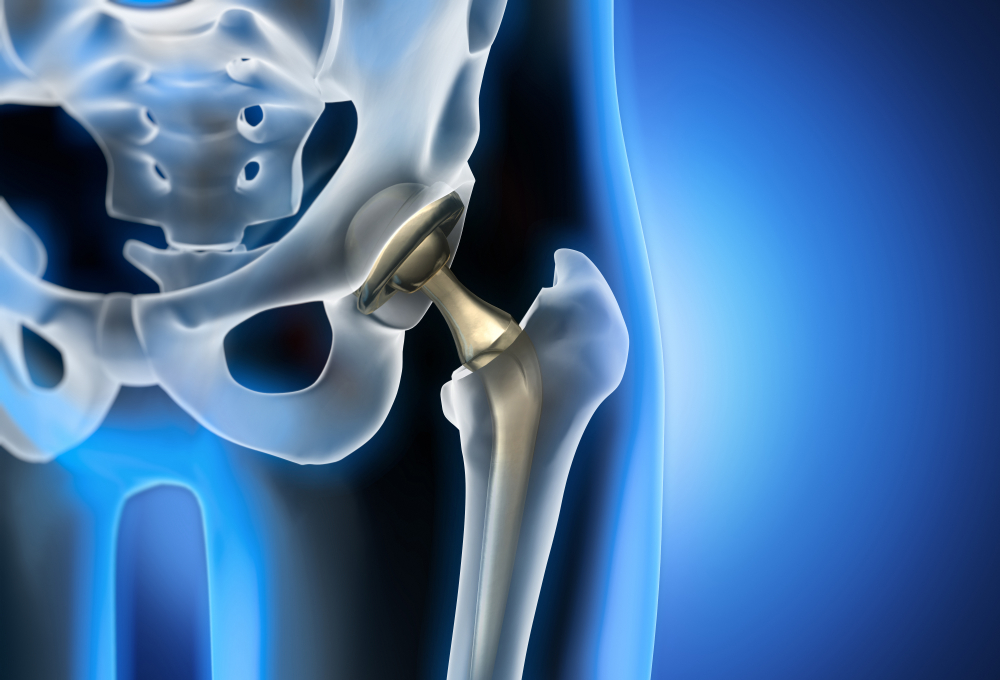
The FDA warns that Synovo Total Hip Systems, implanted after 2019, pose risks of failure and injury. Patients experiencing symptoms should consult our CSS lawyers to explore legal options for compensation.
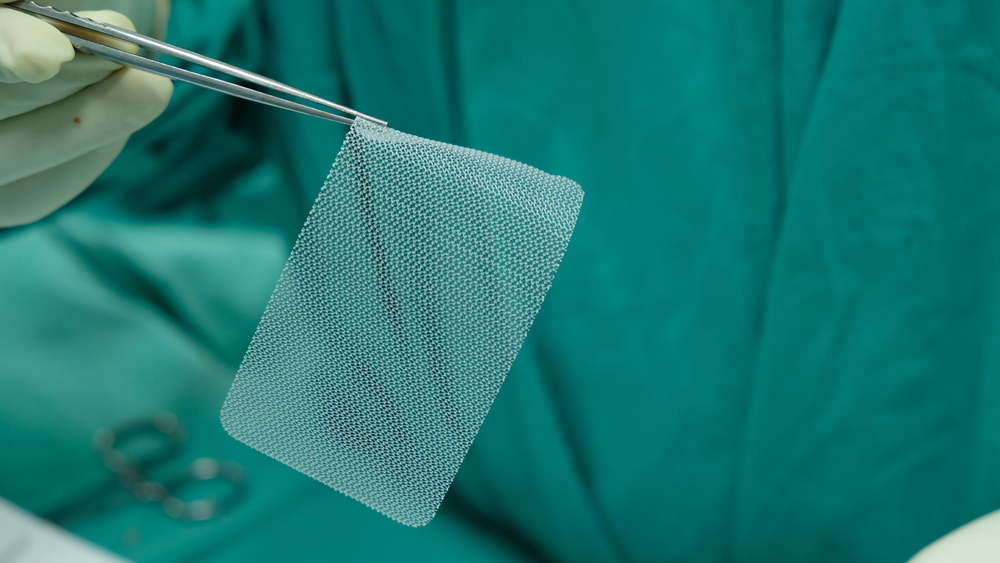
Thousands of hernia mesh products have been recalled, but qualifying for a claim requires proof that a defective hernia mesh implant caused your injury.
Damages from a defective hernia mesh implant vary based on each person’s injuries and circumstances. An experienced dangerous medical device attorney can help identify all the losses you may be entitled to recover.
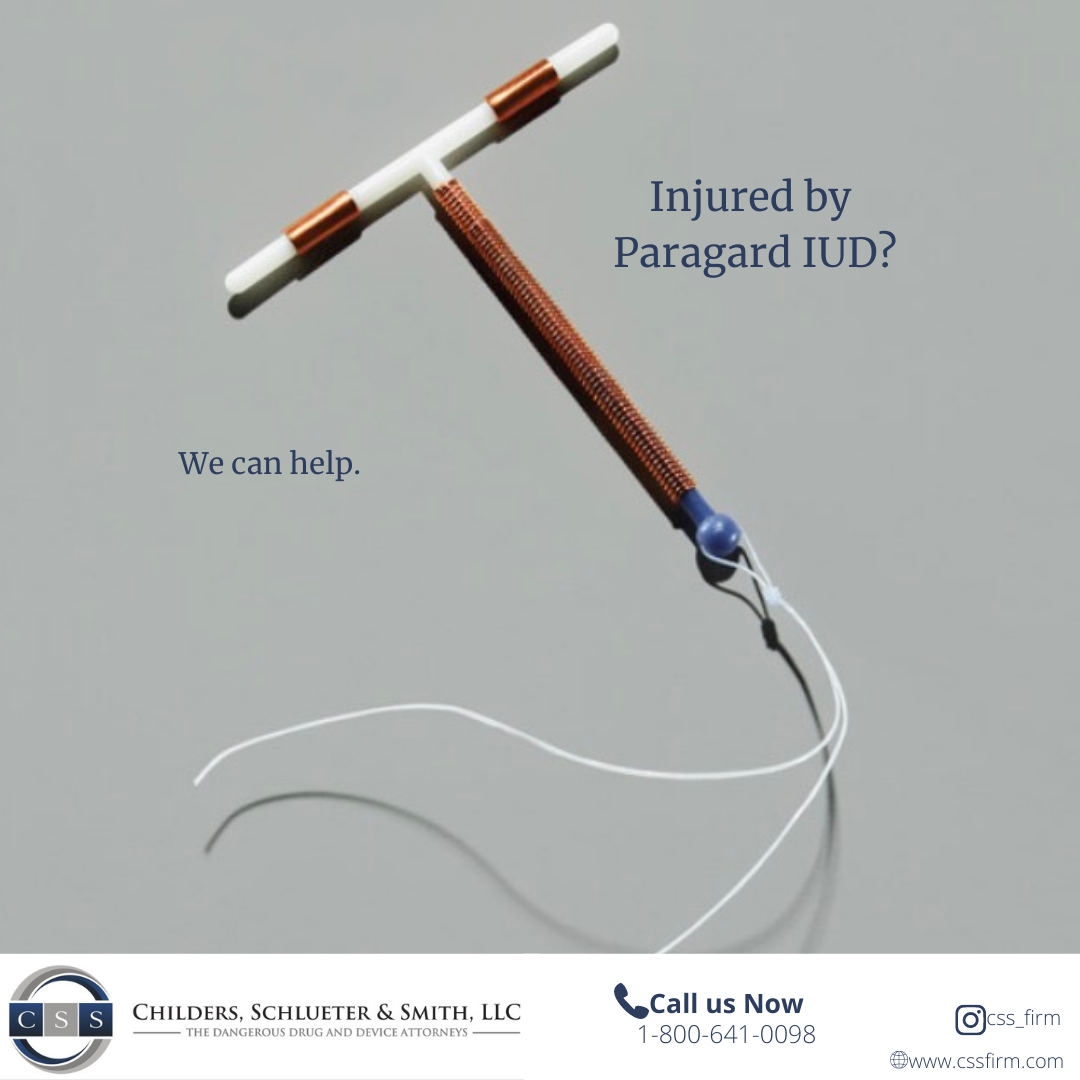
Women nationwide are filing lawsuits after suffering injuries from Paragard IUD breakage—alleging the manufacturer failed to warn of the device’s dangerous design defects.
A voluntary recall affects over 3.5 million Philips Respironics CPAP, BiPAP, and ventilator devices due to foam degradation risks, potentially endangering sleep apnea patients’ health and safety.
Philips Respironics has recalled certain CPAP, BiPAP, and ventilator devices due to degrading sound foam that may release harmful particles and chemicals, posing serious health risks to users.