In a guidance statement released October 30, the U.S. Food and Drug Administration (FDA) made a non-binding recommendation that the makers of ultrasonic surgical aspirators should label them as not recommended for removing uterine fibroids, because doing so could inadvertently spread occult cancer.
In a guidance statement released October 30, the U.S. Food and Drug Administration (FDA) made a non-binding recommendation that the makers of ultrasonic surgical aspirators should label them as not recommended for removing uterine fibroids, because doing so could inadvertently spread occult cancer.
The FDA said that the device’s oscillating tip can disperse the tissue that it breaks up with ultrasonic waves, despite the fact that it is designed to suck up the tissue, and the risk of disseminating cancer cells is outweighed by the aspirator’s benefits, which include “more extensive tumor debulking, little or no collateral thermal damage, and the ability to avoid organ resection,” according to the statement.
Guidance Statement Sounds Familiar
This guidance from the FDA echoes earlier warnings regarding power morcellators and the risks and benefits of laparoscopic fibroid surgery. Like power morcellators, ultrasonic aspirators are devices that break up tissue and then remove it from the body through a small incision. Ultrasonic aspirators are used by a wide variety of surgeons in both open and laparoscopic surgical procedures, including for advanced malignancies that cannot be removed completely. Some such devices are explicitly recommended for uterine fibroid removal, while others have more general uses – for laparoscopic, open and gynecological surgery, which could be interpreted to include fibroid removal, according to the FDA.
It is unclear what prompted the October 30 guidance statement that was first proposed in November 2016, as the FDA said the agency knows of no circumstances when surgeons using ultrasonic aspirators to remove uterine fibroids spreading or upstaging uterine cancer. The American College of Obstetricians and Gynecologists (ACOG) previously agreed that the device was capable of spreading occult uterine cancer but said that the recommendation was “too rigid and eliminates patient choice,” and pointed out that abdominal hysterectomies present greater health risks.
Other Medical Devices News
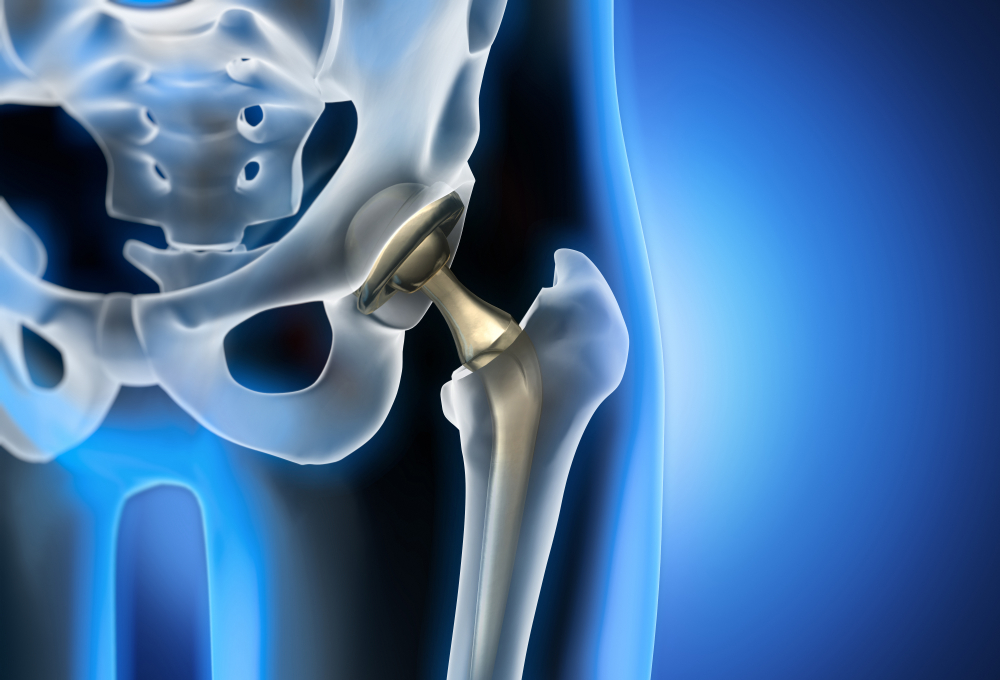
The FDA warns that Synovo Total Hip Systems, implanted after 2019, pose risks of failure and injury. Patients experiencing symptoms should consult our CSS lawyers to explore legal options for compensation.
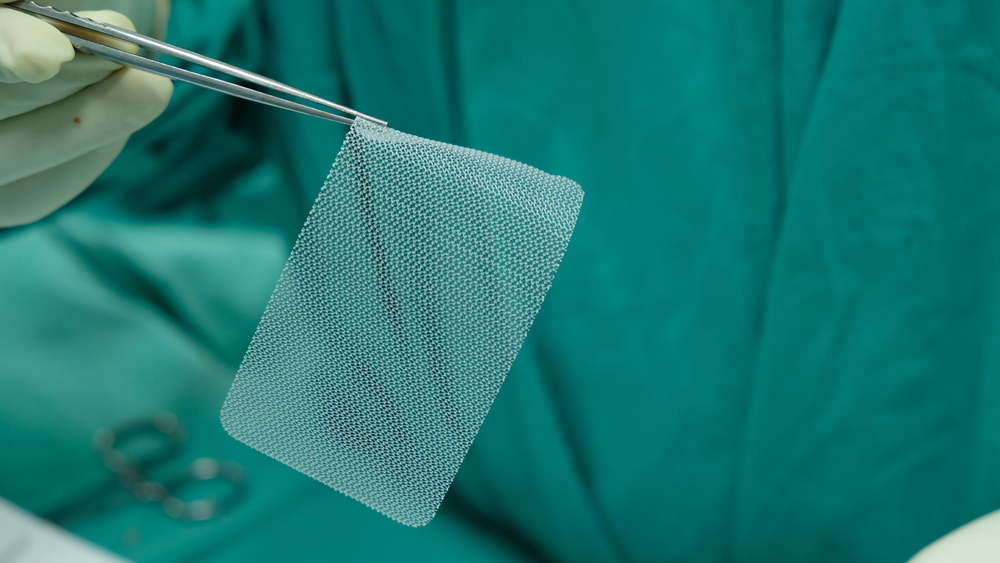
Thousands of hernia mesh products have been recalled, but qualifying for a claim requires proof that a defective hernia mesh implant caused your injury.
Damages from a defective hernia mesh implant vary based on each person’s injuries and circumstances. An experienced dangerous medical device attorney can help identify all the losses you may be entitled to recover.
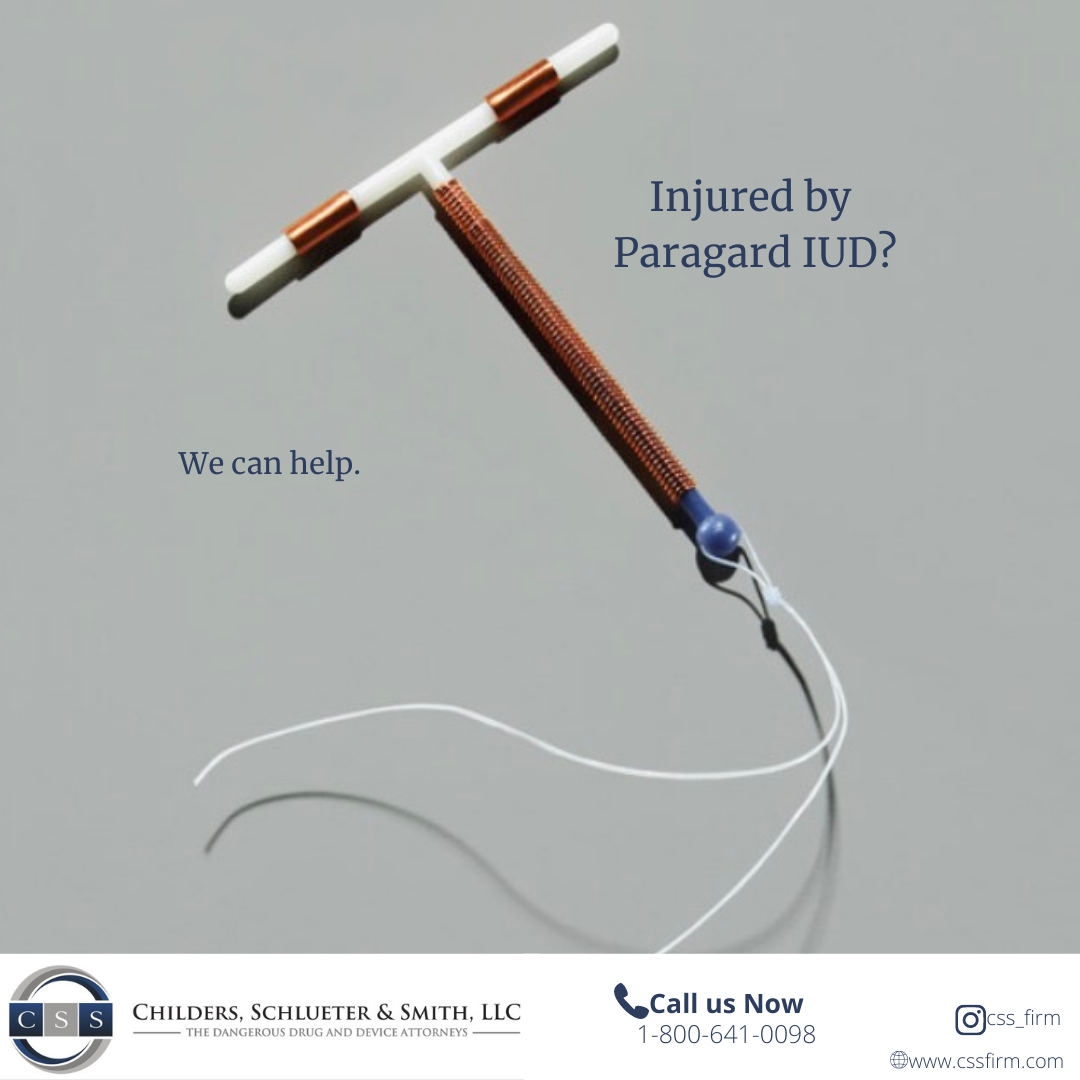
Women nationwide are filing lawsuits after suffering injuries from Paragard IUD breakage—alleging the manufacturer failed to warn of the device’s dangerous design defects.
A voluntary recall affects over 3.5 million Philips Respironics CPAP, BiPAP, and ventilator devices due to foam degradation risks, potentially endangering sleep apnea patients’ health and safety.
Philips Respironics has recalled certain CPAP, BiPAP, and ventilator devices due to degrading sound foam that may release harmful particles and chemicals, posing serious health risks to users.