Please Note: CSS Firm is no longer accepting or investigating new knee implant claims at this time. This article is for educational purposes only.
Knee replacement surgeries are intended to give patients a new knee that is projected to last them decades. Research has concluded that 96 percent of all knee implants are still functional after 10 years, and 80 – 85 percent will last 20 years. However, despite the fact that knee replacement implants are built for long life, some can fail or wear out.
Why Knee Replacements Fail
Knee replacements can fail for many different reasons, such as:
- Incorrect placement of the knee replacement implant.
- Physical factors such as stiffness or weakness in the soft tissue surrounding the knee.
- Infection.
- Fractures due to an injury.
- Wear and tear.
- Unusual degree of friction that leads to bone loss and causes the implant to loosen.
If your knee replacement fails, your body will most certainly let you know, and you will exhibit a variety of symptoms, including pain, swelling, a loss of range of motion in your knee, and stiffness in part of all of the knee.
What to Do if You Suspect Knee Implant Failure:
- Don’t hesitate to contact your surgeon immediately.
- You might also obtain a second opinion from another surgeon.
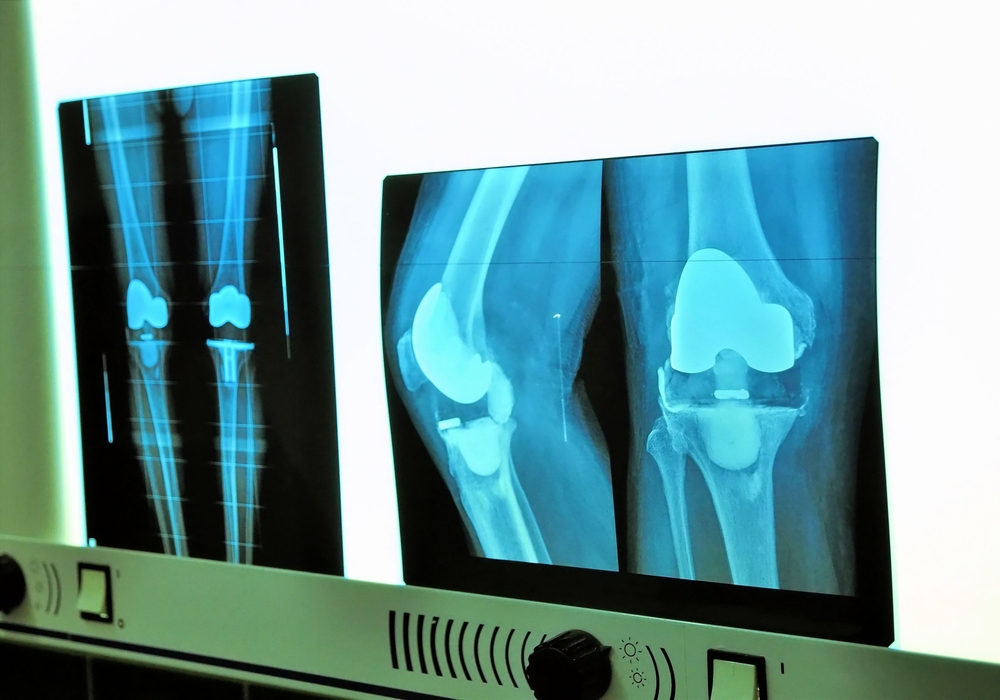
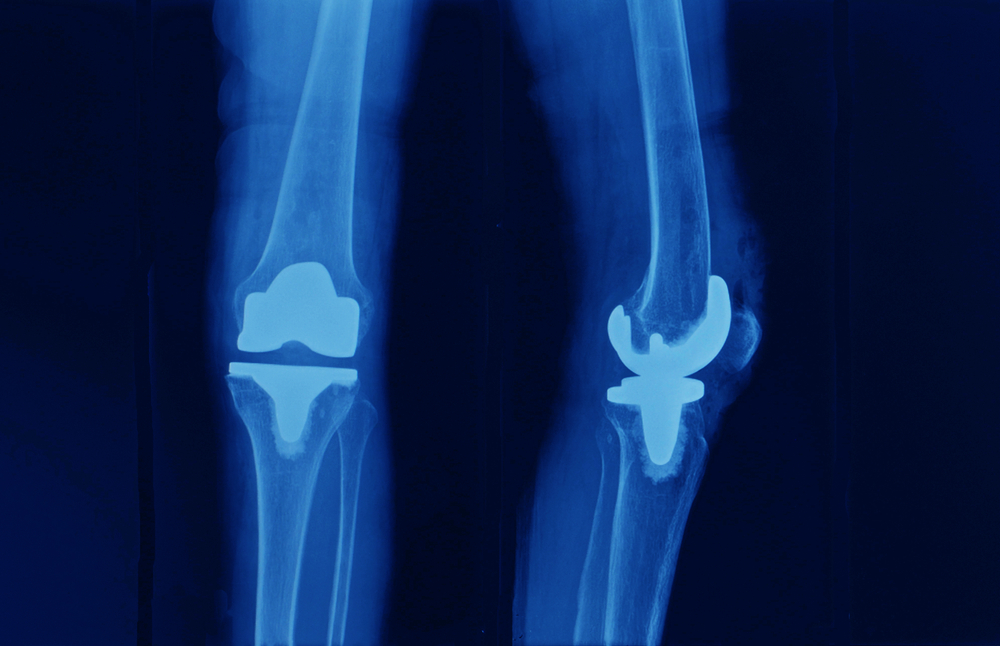
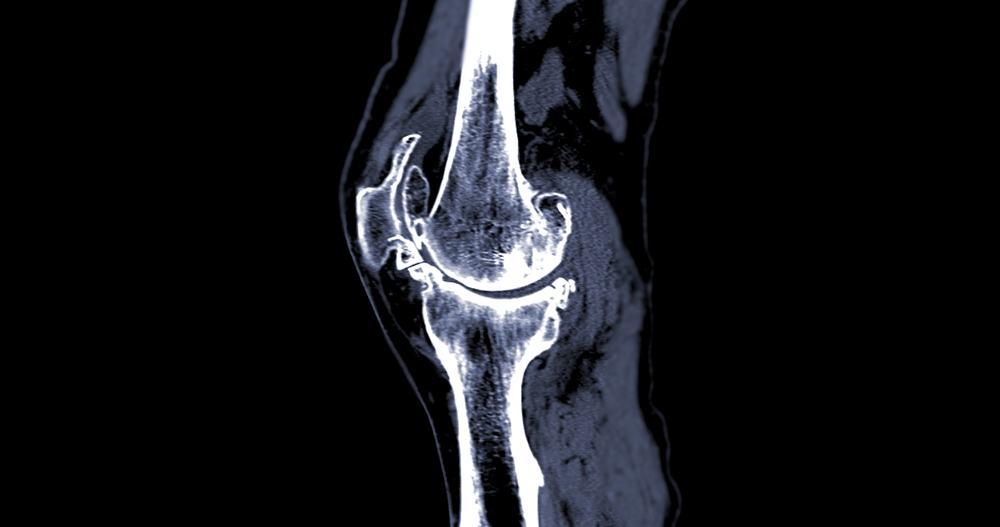
- You will likely be required to undergo a series of tests – X-rays, an MRI, potentially a CT scan, and laboratory tests to diagnose the problem.
If you need revision surgery, your old knee replacement will have to be removed.
If you sustained bone loss, you might require bone grafts, metal wires, screws, and wedges. Revision total knee replacement typically takes longer than an initial knee replacement, and the success rates of revision surgery are usually lower as well.
Lawsuits are currently being filed on behalf of patients who are experiencing problems with their DePuy Synthes Attune Knee Implant system. According to the U.S. Food and Drug Administration (FDA), common issues associated with the Synthes Attune system include failure to bond, worn components, fracture, loss of osseointegration, and the shedding of metal debris.