The Honorable Judge Edmund A. Sargus has been appointed by the United States Judicial Panel on Multi District Litigation (JPML) to oversee pretrial proceedings for the newly centralized Bard hernia mesh lawsuits in MDL 2846
In a transfer order, the JPML ordered all pending Bard Hernia mesh lawsuits be consolidated for discovery and management in the Southern District Court of Ohio. This means 53 lawsuits pending in 21 federal courts are being transferred to 1 court, and will be overseen by District Judge Edmund A. Sargus. The order also mentions 69 potentially related cases that may be transferred. According to the transfer order, these cases are being centralized to “eliminate duplicative discovery, prevent inconsistent pretrial rulings; and conserve the resources of parties, their counsel and the judiciary.”

3D Max Light Hernia Mesh
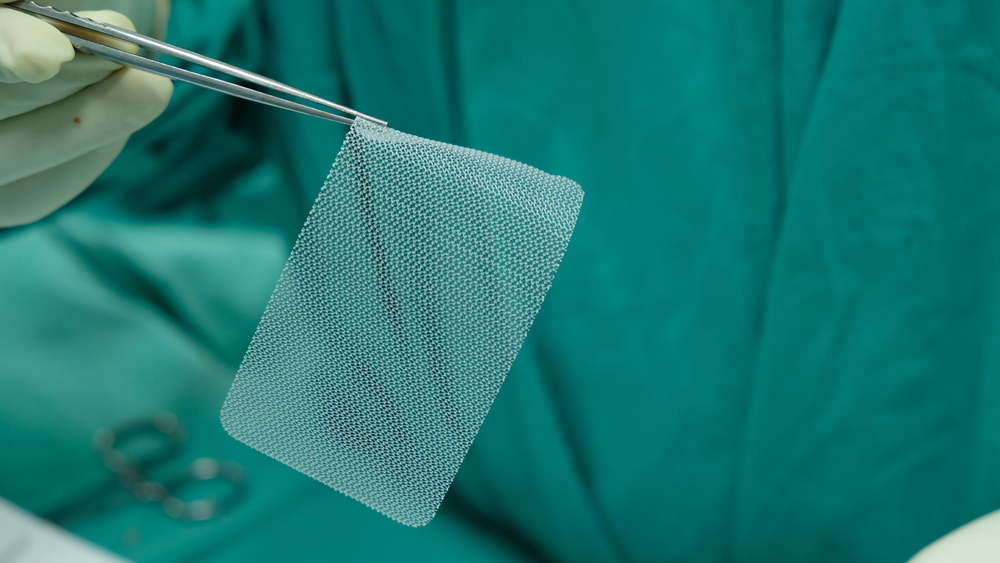
Plaintiffs in these cases allege that the designs of Bard hernia mesh products (polypropylene or plastic hernia patches including Bard Ventralex, Perfex, and Composix) are crucially flawed in ways that can cause severe and potentially life threatening injuries to the patient. These injuries typically result in additional surgeries to either remove or revise the mesh. As more people learn of Bard’s attempt to cover up effects of their faulty mesh products, the hernia mesh litigation is expected to grow substantially over the next few months and years.
“All of the actions share common factual questions arising out of allegations that defects in defendants’ polypropylene hernia mesh products can lead to complications…including adhesions, damage to organs, inflammatory and allergic responses, foreign body rejection, migration of the mesh, and infections.” – “Transfer Order” – Panel on Multi-District Litigation.
“CK Patches” – The JPML further mentions in the transfer order that the defendants’ (Bard and Davol) motion of May 2018 to include the Composix Kugel Hernia Mesh patch would be premature. No CK Patch cases have been brought forth: “We will address this question in due course through the conditional transfer order process.”
Look into the Past: Bard’s Defective TVM Products
This is not the first time Bard is under fire. In the early 2000’s there were tens of thousands of transvaginal mesh (TVM) cases against Bard and a long list of other manufacturers for defective TVM products that also caused severe injury to patients. Multiple mesh companies misled women to believe mesh products were safe and would improve lives. Bard still holds tightly to their claim of safe products. Evidence has consistently shown otherwise; there are many cases, TVM and hernia mesh alike, still pending against Bard today.
As updates develop, we will report back here.